|
|
Post by lowell on Oct 26, 2015 2:13:35 GMT -6
I have dutifully been reading about the beginnings of the new physics. As I promised, I will attempt to represent the high points of what I learned. The book I am reading, "Beyond Einstein" by Michio Kaku, has many charming stories that I will probably skip over and it is a pleasure to read and re-read. What I relate here will be less enjoyable but informative. The unification of the primary forces of nature had its first high point with the theories of a man named Maxwell. For hundreds of years, magnetism and electricity were considered to be two different forces. A thirty year old Scottish physicist named James Clerk Maxwell who studied at Cambridge, claimed in 1860 that electricity and magnetism were not two distinct forces but two sides of the same coin. He had learned that light itself could be better understood by this new way of viewing electricity and magnetism. Maxwell recognized that electric and magnetic fields permeate space and can be viewed as 'arrows' that emanate from an electric charge. Maxwell took it a step further and theorized that it was possible for electric and magnetic fields to vibrate together in precise synchronization so that the wave they create would propagate itself through space. Maxwell envisioned a vibrating magnetic field creating an electric field which in turn vibrated and created yet another magnetic field which in turn created yet another electric field and so on and on. We can think of this as dominoes that are successively triggered to move, but they are alternating black and white dominoes and if the black dominoes are removed the wave can not continue. 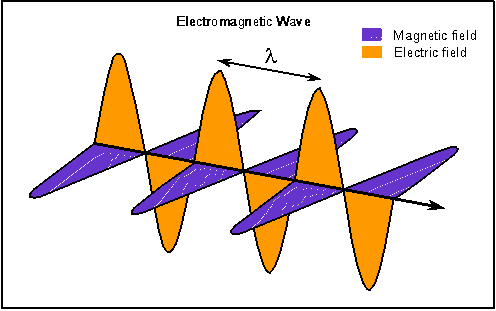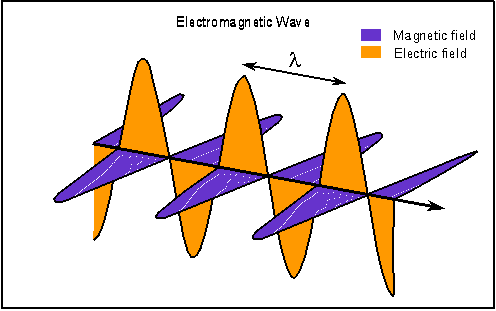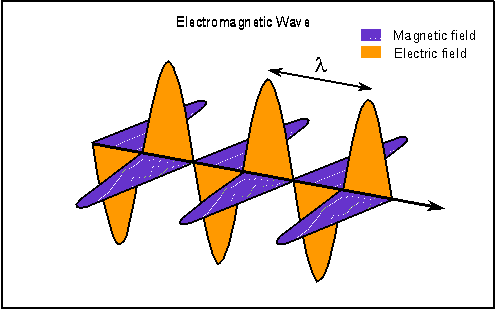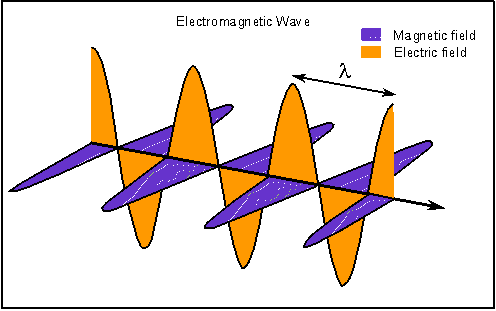 Maxwell used his computations to discover the speed of this wave. To his surprise, it was the speed of light! So Maxwell had discovered the first unification of primary forces, the electromagnetic wave. I will continue with the successive discoveries that lead to the theory of everything. The theory that unifies all the forces of the universe, in replies to this post. |
|
|
|
Post by lowell on Oct 27, 2015 2:47:55 GMT -6
Scores of years later, Einstein gave credit to Maxwell's theory as an influence that developed into the theory of special relativity.
Einstein said, "The theory of relativity owes its origin to Maxwell's equations of the electromagnetic field."
Einstein examines in the theory of general relativity the questions about the largest features of the universe. "Is there a beginning and end to time? "Where is the farthest point in the universe?" "What lies beyond the farthest point?" What happened at the creation?"
|
|
|
|
Post by lowell on Oct 27, 2015 14:32:32 GMT -6
Quantum mechanics had its beginning at approximately the same time as Einstein's theory of relativity. It took the opposite direction of investigation. Heisenberg and associated physicists like Erwin Schrodinger and Niels Bohr, asked "What is the smallest object in the universe? Can matter be divided into smaller and smaller pieces without limit? Their investigation led to discoveries about sub-atomic particles that move much slower than the galaxies and the general theory of relativity is not necessary to understand them.
Merging the two theories has been the quest of physicists ever since the theory of general relativity and the theory of quantum mechanics began.
Quantum mechanics actually had its beginning around the end of the 19th century when physicists could not explain the light given off by "black bodies". For instance if a bar of steel is heated it will glow red and then when heated more will glow white. The theories of the time could not account for this. Their equations indicated that infinite energy should result at high frequency vibration (which is impossible). A German physicist name Max Planck found a solution in 1900. He proposed that rather than being a continuous wave of radiant energy, the energy occurs in definite discreet packets. Planck proposed that physicists had never seen the granular nature of energy because the packets were incredibly tiny. This size is so tiny that we never see quantum effects in daily life. The number that represents the size of these packets is called "Planck's constant".
"Using Planck's strange theory of the quanta, Einstein asked what happens when a particle of light strikes a metal. If light was a particle obeying Planck's theory, then it should bounce the electrons out of some atoms in the metal and generate electricity. Einstein, using Planck's constant, then calculated the energy of the ejected electrons.
It didn't take long for experimental physicists to verify Planck's and Einstein's equations. Planck won the Nobel Prize in 1918 for his quantum theory, followed in 1921 by Einstein for the photoelectric effect."
|
|
|
|
Post by carpathianpeasant on Oct 27, 2015 18:53:04 GMT -6
Keep talking ... even though I don't get most of it.
If electricity and magnetism are two sides of the same coin, one should be negative and one should be positive.
If heated "black bodies" give off light, then the sun, etc., are black bodies at their core ... no?
|
|
orogenicman
RMem
Old enough to remember how to make stone tools
Posts: 189
|
Post by orogenicman on Oct 29, 2015 1:07:54 GMT -6
Keep talking ... even though I don't get most of it. If electricity and magnetism are two sides of the same coin, one should be negative and one should be positive. If heated "black bodies" give off light, then the sun, etc., are black bodies at their core ... no? Magnetism is what you see when the electric field is moving compared to you. Induction is the other side of that coin- When magnetism is moving compared to you, you also see an electric field.
As for the sun, here is an explanation:
physics.stackexchange.com/questions/130209/how-can-it-be-that-the-sun-emits-more-than-a-black-body
The total radiative power emitted by the Sun is equivalent to the total radiative power emitted by an ideal black body with a temperature of 5778 K and a surface area equal to that of the Sun. This 5778 K is the Sun's effective temperature. The spectrum of the Sun is very close to that of a 5778 K black body, but there are deviations. Some are due to absorption and emission, but others result from three key items:
•There is no such thing as black body. The concept of a black body is an idealization based on some simplifying assumptions. The Sun doesn't exactly satisfy those simplifying assumptions.
•That effective temperature of 5778 K is based on total radiative power, the area under the curve of the Planck distribution. If the spectrum of sunlight falls short of the 5778 K black body spectrum some wavelengths it must necessarily rise above the 5778 K black body spectrum at others.
•The primary reason the Sun fails to satisfy the assumptions that underly the Planck distribution is that we are seeing light from multiple temperature sources. The rest of this answer goes into this in detail
The Sun is not a solid body. It doesn't have a surface from which the radiation originates. The radiation we see from the Sun comes primarily from the Sun's photosphere, a roughly 500 kilometer thick layer near the top of the Sun. The chromosphere, transition region, and corona are above the photosphere. While these higher layers do make solar radiation deviate from the ideal black body curve, the primary source is the photosphere itself.
The amount of light that is transmitted into empty space is a sharply increasing function of distance from the center. However, it is not a delta distribution. The light that does get through from those deeper layers has a higher temperature than the layers above it. The bulk of the radiation we see from the Sun comes from a ~500 km thick layer called the photosphere. The top of the photosphere has a temperature of about 4400 K and has a pressure of about 86.8 pascals. The bottom has a temperature of about 6000 K and a pressure of about 12500 pascals.
What we see is a blend of the radiation from throughout the photosphere. Some of the light comes from the top of the photosphere, some from the middle, some from the bottom, roughly weighted by pressure. The total spectrum looks close to that of a 5778 K black body, but the contribution from the bottommost part of the photosphere tilts the spectrum away from the ideal a bit, making the a tiny bit heavy for shorter wavelength radiation.
|
|
|
|
Post by lowell on Oct 29, 2015 4:16:55 GMT -6
The sun is fusing hydrogen into helium. It does this at its core as well. When fusion takes place, energy is released. Some of that energy is in the light spectrum, so there is light at the core of the Sun. That light probably never reaches the surface because of the massiveness of the sun and the many atoms that the light would collide with.
Interestingly enough we are all emitting photons. People emit photons in the long infrared spectrum. A bar of iron (even a cold bar of iron) emits some as well. People in the 19th century probably didn't know that though.
If you are sitting with friends in your living room, some of their photons that were part of their total energy will be absorbed by your body and some of yours will be absorbed by theirs.
It reminds me of a song by Lennon where he says "Well we all shine on, like the moon, and the stars, and the sun."
|
|
|
|
Post by lowell on Oct 29, 2015 5:12:13 GMT -6
In 1923, Louie de Broglie wrote the basic relations that a "matter-wave" should obey, stating that an electron should have a definite frequency and a wavelength just like light waves.
In 1926, Erwin Schrodinger was inspired by the writings of de Broglie. Shrodinger wrote the complete equation (called the Schrodinger wave equation) that these waves should obey. (A different, but equivalent, form of the theory was written by Heisenberg almost simultaneously.) With this the modern theory of quantum mechanics was born.
Before 1926, scientists thought it was hopeless to try to predict the chemical properties of even the simplest compounds. After 1926, however, physicists went from ignorance to almost complete understanding of the equations governing simple atoms. The power of quantum mechanics was so enormous that all of chemistry, could in principle, be reduced to a series of equations.
Quantum mechanics even allows us to calculate the properties of chemicals that we have yet to see in nature.
For example, in a metal, the atoms are arranged in a lattice. The Schrodinger equation predicts that the outer electrons in the metal atoms are bound to the nucleus only loosely and in fact, may roam freely throughout the entire lattice. Even the slightest electric fields can push these electrons around the lattice which in turn creates the electric current. This is why metals conduct electricity. For rubber and plastics, however, the outer electrons are more tightly bound and there are no such free-roaming electrons that can create a current.
Quantum mechanics also led to understanding of semi-conductors, that control the flow of electricity, and so the transistor.
It predicted the laser.
The success of quantum mechanics has altered the foundation of medicine, industry, and commerce. The irony is that quantum mechanics which seems so definitive and clear-cut in its practical applications, actually is based on uncertainties, probabilities, and bizarre ideas.
In 1927, Werner Heisenberg proposed that it is impossible to know the velocity and the location of an object simultaneously. One can never know precisely the position and the velocity of an electron at the same time. This is also a direct consequence of the Schrodinger equation.
According to Heisenberg, this uncertainty arises because, in the subatomic realm, the very act of observing an object changes its position and velocity. In other words, the process of taking a measurement of an atom's system disturbs the system so greatly that it alters its state, making the system qualitatively different from its state before the measurement was taken. For example, an electron is so small that to measure its position in an atom, photons of light must be bounced off of it. However, the light is so powerful that it bumps the electron out of the atom, changing the electron's position and location.
However, one would argue, with a better measuring apparatus, couldn't the velocity and the position of the electron be measured without altering it? According to Heisenberg, the answer is no. Quantum mechanics asserts that we can never know simultaneously, no matter how sensitive our measuring devices are, the exact position and velocity of a single electron. We can know one condition or the other, but not both at the same time. This is called Heisenberg's Uncertainty Principle.
|
|
|
|
Post by lowell on Oct 29, 2015 5:46:50 GMT -6
Newton had the idea that the universe was like a clock that God set in motion at the beginning of time. Newtonian determinism said that the three laws of motion can mathematically determine the precise motion of all particles in the universe.
The French mathematician, Pierre Simon Laplace believed that all future events could be calculated in advance if the initial motion of all of the atoms at the beginning of time were known. For example, determinism in its most extreme form states that it is possible to calculate in advance with mathematical precision which restaurant you will be eating in ten years from now and what you will order.
Moreover, according to this view, whether we wind up in heaven or hell is determined ahead of time. There is no free will.
According to Heisenberg, however, all of this is nonsense. Our fate is not sealed in a quantum heaven or hell. The Uncertainty Principle makes it impossible to predict the precise behavior of individual atoms, let alone the universe. Moreover, according to the theory, in the subatomic realm, only probabilities can be calculated.
We can however, predict with amazing accuracy the probability that a large quantity of electrons will behave in a certain way.
Similarly, in the case of a single radioactive uranium atom, which is unstable and will eventually disintegrate, it can never be predicted precisely when and with what energy it will decay. Without measuring the state of the nucleus, quantum mechanics cannot tell whether it is still intact or whether it has decayed. In fact, the only way quantum mechanics can describe a single nucleus is to assume it is a mixture of these two states.
Before it is measured, a single uranium atom is in a nether state between being intact and decayed. With this strange assumption, quantum mechanics can calculate the rate at which billions of uranium atoms will decay.
Erwin Schrodinger in 1935, devised a clever hypothetical experiment that demonstrates the absurdity of the uncertainty principle.
Imagine a bottle of poison gas, an atom of uranium, and a cat trapped in a box, which we are not allowed to open. Obviously, even though we can't see the cat, we can say that the cat is either alive or dead.
Now imagine that the poison bottle is connected to a geiger counter, which can detect the radiation from a single uranium atom's decay. The apparatus is designed so that if the geiger counter detects a single atom's decay it will release the gas from the bottle and the cat will die.
In this case the cat is described by a wave function that contains the possibilities that the cat is both dead and alive. So the physicist must assume the cat is a mixture of the two states.
Also strangely, it is the act of opening the box that determines which state the cat is in.
Even worse, quantum mechanics implies that objects do not exist in a definite state until they are observed.
Einstein was disturbed by the uncertainty principle, he believed as Newton did, that the physical universe exists in a precise state independent of any measuring process. He said of Schrodinger's cat "No reasonable definition of reality, could be expected to permit this."
|
|
orogenicman
RMem
Old enough to remember how to make stone tools
Posts: 189
|
Post by orogenicman on Oct 29, 2015 7:02:02 GMT -6
The sun is fusing hydrogen into helium. It does this at its core as well. When fusion takes place, energy is released. Some of that energy is in the light spectrum, so there is light at the core of the Sun. That light probably never reaches the surface because of the massiveness of the sun and the many atoms that the light would collide with. Interestingly enough we are all emitting photons. People emit photons in the long infrared spectrum. A bar of iron (even a cold bar of iron) emits some as well. People in the 19th century probably didn't know that though. If you are sitting with friends in your living room, some of their photons that were part of their total energy will be absorbed by your body and some of yours will be absorbed by theirs. It reminds me of a song by Lennon where he says "Well we all shine on, like the moon, and the stars, and the sun." Actually, the light emitted at the sun's core does reach the surface after a 100,000 year journey through its interior. |
|
|
|
Post by lowell on Oct 30, 2015 1:09:33 GMT -6
|
|
|
|
Post by lowell on Nov 2, 2015 6:06:42 GMT -6
Attempts were made to use quantum mechanics to explain the observable behavior of galaxies but because the speed of those was closer to the speed of light the math failed to work.
"For instance, in high school we learn that force fields such as gravity abnd the electrid field obey the "inverse square law" - that is, the farther one distances oneself from a particle, the weaker the field becomes. The farther one travels from the sun, for example, the weaker its gravitational pull will be. This means, however, that as one approaches the particle, the force rises dramatically. In fact, at its surface the force field of a point particle must be the inverse of zero squared which is 1 divided by zero.
Expressions such as 1/0, however, are infinite and ill-defined. The price we pay for introducing point particles into our theory is that all our calculations of physical quantities, such as energy, are riddled with 1/0s. This is enough to render a theory useless, calculations with a theroy plagued with infinities cannot be made because the results cannot be trusted."
"The problem of infinities would haunt physicists for the next half century. Only with the advent of the superstring theory has the problem been solved, because superstrings banish point particles and replace them with a string. "
|
|
|
|
Post by lowell on Nov 2, 2015 6:21:48 GMT -6
"Richard Feynman was an accomplished safe-cracker who broke into some of the most closely guarded safes in the world He also was a world-renowned physicist According to Feynman, both the safe-cracker and the physicist are adept at sifting through seemingly random clues and piecing together subtle patterns that hold the answers to the problem.
Since the 1930s, physicists have been consumed by the frustrating task of cracking the "safe" of quantum field theory and finding the key to the successful marriage between quantum mechanics and relativity. Only in the past twenty years, however, have physicists realized that the tantalizing clues found in the experimental data from atom smashers form a systematic pattern."
"Today we realize that this pattern can be expressed as an underlying mathematical symmetry that links forces that appear to be totally dissimilar. These symmetries, we shall see, will play a central role in canceling the divergences found in quantum field theory. The discovery that these symmetries can cancel these divergences is perhaps the greatest lesson of the past half century in physics"
|
|
|
|
Post by lowell on Nov 3, 2015 6:14:23 GMT -6
'This knack of exploiting symmetries and isolating the key factors in any problem led Feynman to produce the first successful union of special relativity with quantum electrodynamics in 1949, for which he and his colleagues won the Nobel Prize in 1965.
The theory, called "quantum electrodynamics (QED)," was a modest contribution by today's standards, treating only the interactions of the photon (light) and the electron (and not the weak or nuclear force and certainly not gravity); but it marked the first major advance, after years of frustration, in uniting special relativity with quantum mechanics.
The QED theory was as different from relativity as Feynman's personality was different from Einstein's. Unlike most other physicists, Einstein had a playful streak and took every opportunity to poke fun at the stuffy totems of conventional society. But if Einstein was playful, physicist Richard Feynman was an outlandish prankster.
Feynman's early fascination with practical jokes surfaced while he was a young physicist working on the atomic bomb project in the 1940's Priding himself on his abilities as a safecracker, one day he cracked three vaults in a row at Los Alamos that contained the sensitive military equations of the atomic bomb. In one vault he left a message scribbled on yellow note paper bragging how easy it was to crack open the safe, and signed the message "Wise Guy." In the last vault, he put in a similar message and signed it "Same Guy".
|
|
|
|
Post by lowell on Nov 3, 2015 16:24:24 GMT -6
'The next day, Dr. Frederic de Hoffman opened the safes and found these mysterious messages lying atop the most closely guarded secrets in the world. Feynman recalled: "I had read in books that when someone is afraid, his face gets sallow, but I had never seen it before. Well, it's absolutely true. His face turned gray, yellow green - it was really frightening to see." Dr. de Hoffman read the sheet signed by the mysterious "Same Guy" and immediately yelled, "It's the same guy who's been trying to get into Building Omega!" In his hysteria, Dr. de Hoffman falsely concluded that the safecracker was the same man who was apparently spying on another top-secret project at Los Alamos. Feynman soon confessed to being the culprit.
When Feynman was a student at the Massachusetts Institute of Technology, he asked himself a simple question: What is the most important problem in all of theoretical physics? Clearly, it was the elimination of the infinities infesting the quantum field theory.
Feynman set out to predict numerically what happened when particles, such as electrons or atoms, bumped into one another. When describing such collisions, physicists use the term S-matrix (s stands for "scattering), which is merely a set of numbers that contains all the information of what happens when particles collide. It tells us how many particles will scatter at a certain angle with a certain amount of energy.
Calculating the S-matrix is profoundly important because if the S-matrix were known completely, in principle it would be possible to predict virtually all the properties of the material.
One importance of the S-matrix is that it can explain puzzling, everyday phenomena. For example, physicists in the nineteenth century, using a crude form of the S-matrix for the scattering of sunlight in the air, were able to explain for the first time why the sky was blue and sunsets were red.
When we look at the sky during the daytime, we mainly see sunlight that has bounced off air molecules and scattered in all direction in the atmosphere. Because blue light scatters more easily than red light, and because the light from the sky is mostly scattered light, the sky appears blue. (If, however, we imagined a world without air, the sky would look dark even in daylight, because there would be no scattered light. On the moon, which has no air to scatter the sunlight, the sky appears black, even in the daytime.)
The sunset looks red, however, due to the opposite effect: We are looking mainly at the sun itself and not at scattered light. At sunset, the sun sits near the horizon, so light from the setting sun must travel horizontally to reach our eyes and thus travels through a relatively large amount of air. By the time sunlight reaches us, only the reds are left unscattered.
|
|
Deleted
Deleted Member
Posts: 0
|
Post by Deleted on Nov 4, 2015 10:25:08 GMT -6
Keep talking ... even though I don't get most of it. If electricity and magnetism are two sides of the same coin, one should be negative and one should be positive. If heated "black bodies" give off light, then the sun, etc., are black bodies at their core ... no? Magnetism is what you see when the electric field is moving compared to you. Induction is the other side of that coin- When magnetism is moving compared to you, you also see an electric field.
As for the sun, here is an explanation:
physics.stackexchange.com/questions/130209/how-can-it-be-that-the-sun-emits-more-than-a-black-body
The total radiative power emitted by the Sun is equivalent to the total radiative power emitted by an ideal black body with a temperature of 5778 K and a surface area equal to that of the Sun. This 5778 K is the Sun's effective temperature. The spectrum of the Sun is very close to that of a 5778 K black body, but there are deviations. Some are due to absorption and emission, but others result from three key items:
•There is no such thing as black body. The concept of a black body is an idealization based on some simplifying assumptions. The Sun doesn't exactly satisfy those simplifying assumptions.
•That effective temperature of 5778 K is based on total radiative power, the area under the curve of the Planck distribution. If the spectrum of sunlight falls short of the 5778 K black body spectrum some wavelengths it must necessarily rise above the 5778 K black body spectrum at others.
•The primary reason the Sun fails to satisfy the assumptions that underly the Planck distribution is that we are seeing light from multiple temperature sources. The rest of this answer goes into this in detail
The Sun is not a solid body. It doesn't have a surface from which the radiation originates. The radiation we see from the Sun comes primarily from the Sun's photosphere, a roughly 500 kilometer thick layer near the top of the Sun. The chromosphere, transition region, and corona are above the photosphere. While these higher layers do make solar radiation deviate from the ideal black body curve, the primary source is the photosphere itself.
The amount of light that is transmitted into empty space is a sharply increasing function of distance from the center. However, it is not a delta distribution. The light that does get through from those deeper layers has a higher temperature than the layers above it. The bulk of the radiation we see from the Sun comes from a ~500 km thick layer called the photosphere. The top of the photosphere has a temperature of about 4400 K and has a pressure of about 86.8 pascals. The bottom has a temperature of about 6000 K and a pressure of about 12500 pascals.
What we see is a blend of the radiation from throughout the photosphere. Some of the light comes from the top of the photosphere, some from the middle, some from the bottom, roughly weighted by pressure. The total spectrum looks close to that of a 5778 K black body, but the contribution from the bottommost part of the photosphere tilts the spectrum away from the ideal a bit, making the a tiny bit heavy for shorter wavelength radiation.
Why does it keep repeating it's self? There are so many stars it would seem it would be a relatively common accourrence. |
|



